New viruses continue to emerge and spread. Joan B Rose, Rosina Girones, Marta Rusiñol Arantegui, Veronica Beatriz Rajal, Ramiro Poma, Maronel Steyn, Bettina Genthe, Susan Petterson, and Nicholas Ashbolt explain how water professionals can monitor, treat and help decision-makers respond to the spread of viral diseases.
Since the pandemic, viruses have featured regularly in the news – be it COVID-19, monkeypox or the recent rise in polio cases in parts of the world where the disease was previously considered eradicated. Increasingly, the value of wastewater-based epidemiology (WBE) is becoming recognised as a tool to support public health, helping to track the spread of disease across populations, prevent the spread of transmission and inform vaccination programmes.
Traditionally, when examining the occurrence of faecal-oral pathogens in wastewater, the focus has been on downstream efforts for the treatment, removal and disinfection of microorganisms that have the potential for transmission through waterborne infection. However, with the impact of COVID-19, the development of upstream applications for monitoring the spread of the disease within communities increased rapidly. Many countries have successfully implemented wastewater monitoring to track excreted SARS-CoV-2 and its variants using open access platforms and international initiatives.
Since 2019, several variants of concern (VOC) have emerged, including different subvariants. A VOC is identified by the World Health Organization (WHO) and Centers for Disease Control and Prevention (CDC) as a variant that is more transmissible, spreading globally and/or causing more serious illness. Table 1 (below) shows the most common global variants and the date and location they were first observed.
The VOCs have all been identified in wastewater via targeted polymerase chain reaction (PCR) assays and/or sequencing. It has been shown that there is a large variability of viral load in excreta over time of infection and between patients, and it is presumed that differences also exist among the array of variants.
 The most recent VOCs are the Omicron sub-lineages BA.4 and BA.5, with more than 50 mutations, mainly located in the spike protein. Both BA.4 and BA.5 variants have mutations that are linked to immune system evasion and antiviral treatment resistance. The recently observed increase in the circulation of sub-lineages BQ.1, which originated from BA.5, is probably driven mainly by immune escape. This variant or other omicron sub-lineages probably contributed to the further increase in cases of COVID-19. However, it is unlikely that critical cases will oversaturate the health systems, as was observed during the first waves. This can be attributed to increased immunity because of vaccination programmes and natural immunity from previous infections.
The most recent VOCs are the Omicron sub-lineages BA.4 and BA.5, with more than 50 mutations, mainly located in the spike protein. Both BA.4 and BA.5 variants have mutations that are linked to immune system evasion and antiviral treatment resistance. The recently observed increase in the circulation of sub-lineages BQ.1, which originated from BA.5, is probably driven mainly by immune escape. This variant or other omicron sub-lineages probably contributed to the further increase in cases of COVID-19. However, it is unlikely that critical cases will oversaturate the health systems, as was observed during the first waves. This can be attributed to increased immunity because of vaccination programmes and natural immunity from previous infections.
Hepatitis of unknown etiology
Numerous cases of liver inflammation among children have been reported in the world this year. The alert was first raised by the UK Health Security Agency (UKHSA) on 5 April 2022, notifying 10 cases of severe acute hepatitis of unknown etiology in previously healthy young children under 10 years old. By 22 June 2022, 920 probable cases (fitting the WHO case definition) from 33 countries were reported to WHO, including some retrospectively identified from 2021. Forty-five children required liver transplants (5%) and 18 (2%) deaths were registered by WHO in 2022.
Around half of the reported probable cases were in Europe (460 cases), where the UK accounted for 58% of them. This was followed by the Americas (383 cases, 80% from the USA), Western Pacific Region (61 cases), South-East Asia Region (14 cases), and Eastern Mediterranean Region (two cases). In 422 cases of the total, where gender and age were registered, 52% were female and 78% were under six years old.
Since the beginning of the alert, researchers have been trying to identify the etiological agent of the mysterious cases of hepatitis, without success to date. However, there are two different theories. The Centers for Disease Control and Prevention in the USA has explored the possible connection to inflammation with SARS-CoV-2, as some of the children with hepatitis tested positive for COVID-19 before the liver illness. The UKHSA’s main hypothesis is that this hepatitis is caused by an adenovirus, as many children with the mysterious hepatitis in the UK and USA were also infected with adenovirus, although with low viral titers. In two other studies, adenovirus was present in only a small proportion of children with liver inflammation; however, all the children had previously tested positive for SARS-CoV-2 or had family members who had. A retrospective case study reported five children who presented two different patterns of potentially long COVID-19 liver injury, after discarding other known etiologies. However, the connection of SARS-CoV-2 infection with these cases of hepatitis has recently waned.
 Adenovirus is now the most frequently detected pathogen among cases with available information, detected in 203 of 371 cases (55%) in Europe, and 113 of 252 cases (45%) in the USA. In 327 cases out of 920, the children were found to be positive for adenovirus by PCR; of these, 44 (13.5%) were identified as being positive for adenovirus type 41, which is generally associated with gastrointestinal disease. According to the viral immunohistochemistry results of the liver samples’ intrasinusoidal lumen, this condition may also be linked to immunologic damage brought on by adenovirus.
Adenovirus is now the most frequently detected pathogen among cases with available information, detected in 203 of 371 cases (55%) in Europe, and 113 of 252 cases (45%) in the USA. In 327 cases out of 920, the children were found to be positive for adenovirus by PCR; of these, 44 (13.5%) were identified as being positive for adenovirus type 41, which is generally associated with gastrointestinal disease. According to the viral immunohistochemistry results of the liver samples’ intrasinusoidal lumen, this condition may also be linked to immunologic damage brought on by adenovirus.
In various surveys around the world, more than 80% of sewage has been found positive for adenovirus. The concentrations of adenovirus in sewage have been found to range from 104 to 109 gene copies/litre and are often found in secondary (treated) effluent. The potential association of adenovirus with hepatitis in children adds further importance to sewage containment and treatment.
Monkeypox
Monkeypox is a viral zoonotic disease first identified in 1958 in monkeys kept in captivity for research purposes in Denmark, even though in nature it is most often found in rodents. The first human case was discovered in 1970 and, since then, it has been endemic to the tropical rainforest areas of Central and West Africa. More recently, the disease has been found in urban areas and the first outbreak outside of Africa was in 2003, in the USA (imported from Ghana through infected animals).
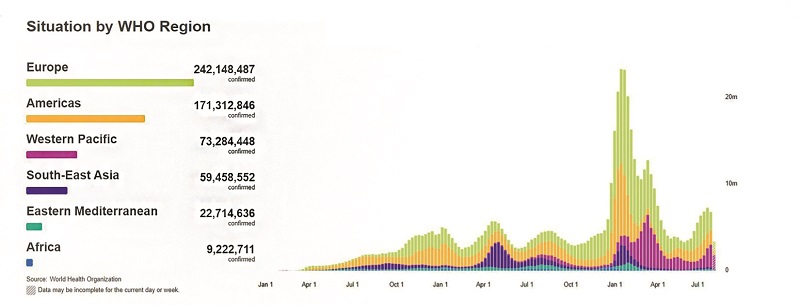
The current monkeypox outbreak was reported on 7 May 2022 in the UK, and continued to spread to 110 countries (of which only seven countries in Africa had previously reported monkeypox) and infected more than 80,634 people globally. To date, 55 deaths were confirmed. Until this outbreak, monkeypox cases in people outside of Africa were predominantly linked to international travel to countries where the disease commonly occurs, or through imported animals.
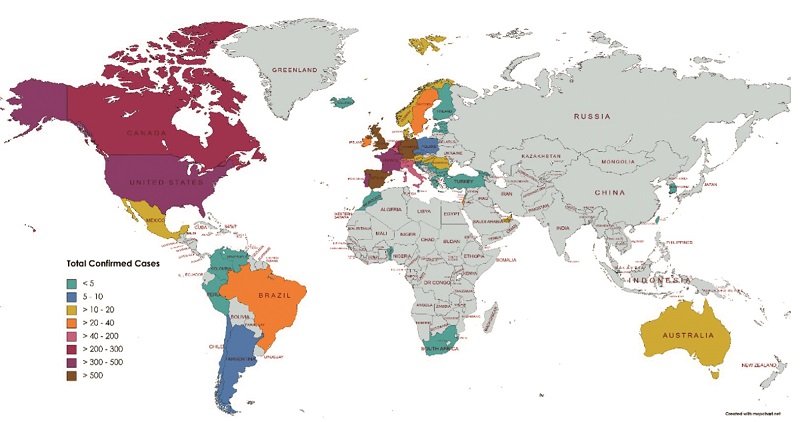
Global cases of monkeypox
Genome analysis indicates that the milder West African strain is responsible for the current outbreak. Interestingly, however, most of the current cases seem to have occurred as a result of person-to-person transmission, which has rarely been seen before.
The virus has been detected in saliva, rectal swabs, urine and faecal samples, and has the potential to be released from the skin into greywater when infected individuals wash. While, initially, monkeypox virus and its DNA had not been detected in wastewater or biosolids, the first confirmation of monkeypox by PCR was recently made in two samples of wastewater, including in primary solids in California. Further studies are needed to determine if wastewater-based surveillance monitoring techniques, similar to that used during the COVID-19 outbreak, could or should be used to monitor monkeypox.
Provision of sanitation systems and the treatment and disinfection of faecal wastes and wastewater have been major engineering feats of the 20th century, reducing infant mortality and improving public health generally. When a new pathogen emerges, primary considerations for the water industry are:
- Does this agent occur in bodily fluids and in wastewater, and how is it transmitted?
- How long does the agent remain infectious in water systems?
- How effective are our existing sanitation and water systems for managing this new agent?
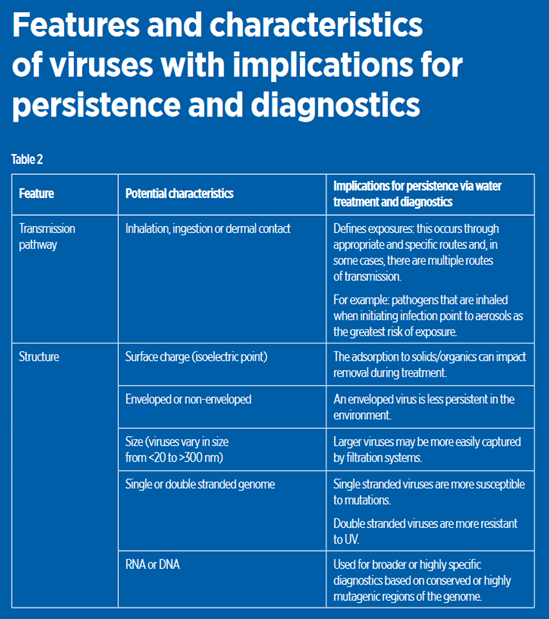
New technology and the rapid sharing of information make it possible now to answer these questions for any apparently new viral agent that may be detected in wastewater. Table 2 (over) summarises some key features of viruses that provide useful starting points, as these characteristics and the rapid information on the water microbiome and genomes of key pathogens in water allow for transformational knowledge on any new agent found in wastewater.
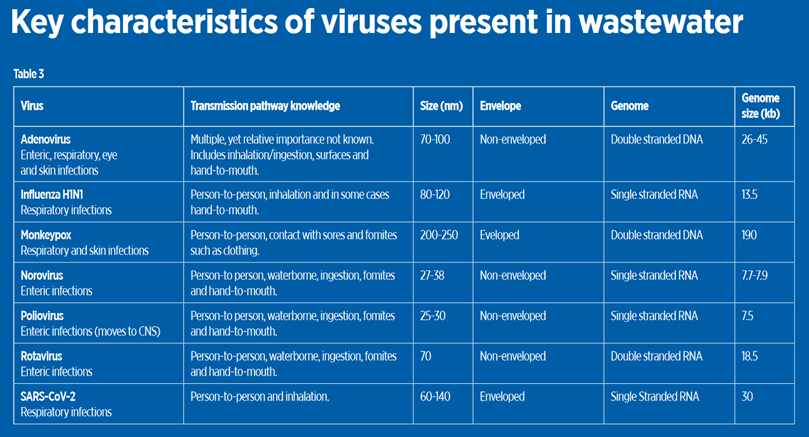
Table 3 presents some of the key viruses of interest that could be monitored in wastewater for assessing community disease, mutations/variants and wastewater treatment efficacy. Some may be found at consistently high levels in sewage because of replication in the intestinal tract (including the enteric viruses and SARS-CoV-2), while others enter wastewater indirectly (for example, monkeypox and influenza) coming from skin, wounds or sputum, but without replication in the intestinal tract. Data on occurrence, persistence, removal, and inactivation is summarised in Sanitation and Disease in the 21st Century, which is an online resource (www.waterpathogens.org/book/section-i-viruses).
Environmental persistence has always been one of the key characteristics of waterborne viral pathogens. The relative persistence of human infectious viruses is generally considered to be highest for non-enveloped virions, such as the non-enveloped human noroviruses, the most common cause of gastroenteritis. So, if the water industry can manage norovirus exposures, it is likely to be able to manage less numerous and more fragile emerging viruses. Many emerging viruses are enveloped, which when freely suspended in environmental waters are inactivated faster (e.g., time for 90% inactivation (T-90) for SARS-CoV-2 in sewage is 1.5 days, compared with the enteric viruses (e.g., adenoviruses with a T-99 of >40 days). There is also evidence for even greater persistence when consumed by free-living protozoan grazers, common in sewage and treatment works, which surprisingly may not digest various virus particles but, indeed, excrete parcels of infectious viruses within membrane vesicles that offer 90-99% protection from UV and chemical inactivation, and even higher protection when they end up in their host’s cysts, providing further reason to manage droplet/aerosols from wastewaters.
This was well illustrated by the case of SARS-CoV-1. The early expectation from observed transmission pathways (person-to-person via inhalation), combined with the knowledge that coronaviruses are enveloped (shorter environmental survival), meant that transmission via the sewer network was not expected to present a significant risk.
Nevertheless, investigation of the outbreak in the Amoy Gardens high-rise complex in Hong Kong in 2003 implicated the sanitation system. Sewage droplets travelled at least 200m to provide infectious doses of the SARS-CoV-1 virus via inhalation. This highlighted the need to minimise aerosol generation at treatment works, not only for local workers, but communities downwind of treatment works too.
Currently, it is believed that most emerging viruses have zoonotic origins, thus primary management of human exposures to emerging viruses via water now resides with those responsible for handling animal wastes and the areas of interface with native fauna, such as live meat markets and agroforestry. When infections arise in communities, it is the water industry’s responsibility to consider how treatment systems can provide controls to reduce novel viral pathogens and how wastewater monitoring can help to inform public health decisions.
The wastewater industry can play a significant role in treating sewage, thereby reducing risks from waterborne diseases, and in the equitable distribution of monitoring programmes. WBE can inform us of the evolution of SARS-CoV-2 in a population and has been extremely useful for the surveillance of novel pathogens and the emergence or re-emergence of new viral strains. The most successful strategy to mitigate the spread of emerging or re-emerging viruses is to strengthen global health systems. WBE has an important part to play in this and can support the equitable distribution of vaccines for population-wide immunity. We are not there yet, however, and it remains prudent to implement general infection prevention and control practices, including appropriate wastewater treatment for all well-known enteric viruses. The water sector must begin working with laboratories around the world to build a community of practice for monitoring viral and other pathogens in wastewater to support public health measures.
The authors:
Joan B Rose is Homer Nowlin endowed chair in water research at Michigan State University, USA;
Rosina Girones is research group leader at the Departament de Genètica, Microbiologia i Estadística Universitat de Barcelona, Spain;
Marta Rusiñol is researcher at the Departament de Genètica, Microbiologia i Estadística, Universitat de Barcelona, Spain;
Veronica Beatriz Rajal is researcher at Instituto de Investigaciones para la Industria Química (INIQUI) and the Facultad de Ingeniería, Universidad Nacional de Salta – Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), Argentina;
Hugo Ramiro Poma is researcher at the Instituto de Investigaciones para la Industria Química (INIQUI), Universidad Nacional de Salta – CONICET, Argentina;
Maronel Steyn is senior researcher at the Council for Scientific and Industrial Research, Stellenbosch, South Africa;
Bettina Genthe is senior researcher at the Council for Scientific and Industrial Research, Stellenbosch, South Africa;
Susan Petterson is associate professor at the School of Medicine and Dentistry, Griffith University, Australia;
Nicholas Ashbolt is executive dean at the Faculty of Science and Engineering, Southern Cross University, Australia.
More information
www.who.int/news/item/22-06-2022-vaccine-derived-poliovirus-type-2-(vdpv2)-detected-in-environmental-samples-in-london–uk
Contact rosejo@msu.edu for the complete list of references for this article.